Posted: May 2023
NewsWe’re proud to announce that NRL was awarded USD $1.2 million to provide a Quality Assurance Program to support COVID-19 Rapid Diagnostic Testing (RDT) at Point of Care (POC).
The funding comes from the Foundation for Innovative New Diagnostics (FIND), overseeing the Diagnostic Pillar of the Access to COVID-19 Tools Accelerator (ACT-A) partnership, hosted by the World Health Organisation (WHO).
The ACT-A partnership recognised that the SARS-CoV-2 pandemic could not be controlled unless everyone, especially those living in low- and middle-income countries, (LMIC) could access COVID-19 healthcare support.

-
Components of the project
This project was composed of three components:
- Calibration of a sample to a certified reference material be used in the manufacture of Quality Assurance Products
- Enrolment of community-based PoC test sites from a range of LMICs into the QA program
- Development of a QA data submission portal using smartphone technology to provide test sites with real-time troubleshooting support
Results received by country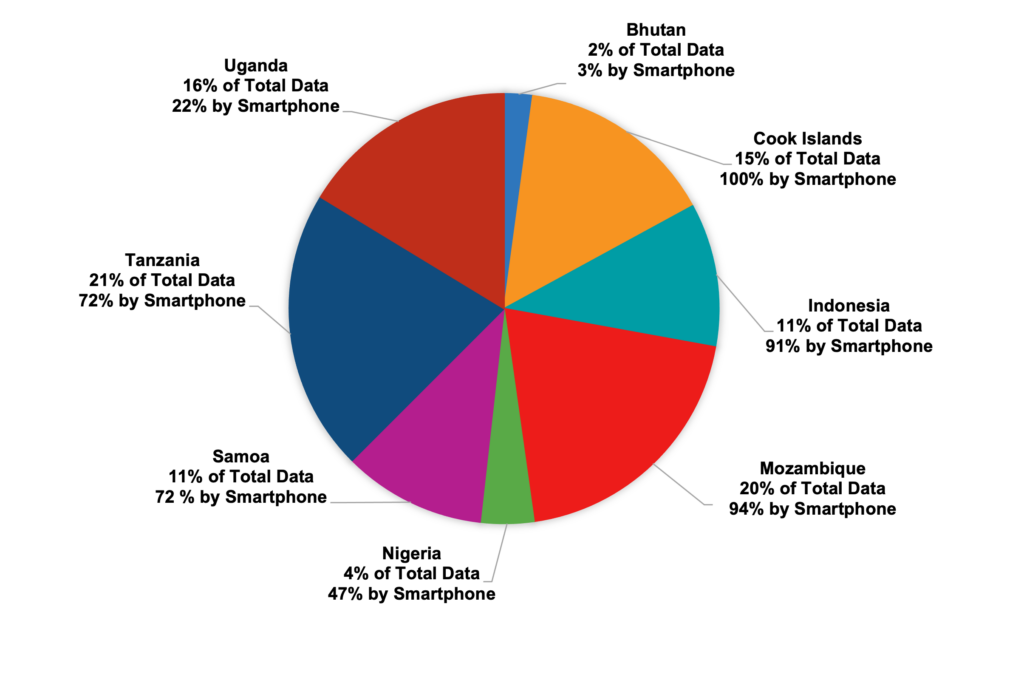
Fig.1: Results received by country. As of May 1st, 2023, 2267 results were received from 93 POC test sites using 14 different COVID-19 RDTs
Conclusions & ImpactData review will help us understand whether sample manufacture, sample and RDT shipping & storage, operator proficiency, RDT test performance or a combination of these affect the quality of test results reported at the POC test sites.
NRL continues to support these countries while they still maintain stocks of QA materials. Meanwhile, the QA method implemented by NRL to these countries could be applied to other infectious diseases that benefit from high quality decentralised, POC-based diagnostic testing such as HIV, hepatitis, and Tuberculosis.